
Over time this gas and dust became part of clouds that formed new stars, which also fused hydrogen and helium into heavier elements until they too died in supernova explosions. The gas and dust remnants of these stars were tossed out into the universe. After several hundred thousand years these first stars had no further way to produce energy, and in the end they explode in a massive explosion known as a supernova. Beyond iron there are no elements a star can fuse to produce energy. Toward the end of a their lives they produced even heavier elements, such as silicon, neon, and eventually iron. The first stars are thought to have been very large stars. As a result, these elements become fairly plentiful within a star. As a star ages the CNO cycle becomes the dominant process by which a star creates light and heat.

The carbon interacts with the hydrogen to produce nitrogen and oxygen as well as helium, through a process known as the CNO cycle. As helium become more plentiful in the stars core, some of it fuses into carbon. The energy produced by this nuclear fusion gives a star the light and heat necessary to counter the force of gravity for a time, but as a star ages the amount of helium in the stellar core increased. They formed from large clouds of hydrogen and helium, and as they collapsed under their own weight the hydrogen in their cores began to fuse into helium. For the next several hundred million years only these four elements existed. The elements produced by the big bang consisted of about 75% hydrogen and 25% helium (by mass) with trace amounts of lithium and beryllium. The first elements appeared a few minutes after the big bang, through a process known as nucleosynthesis. To understand why, we have to look to the stars.
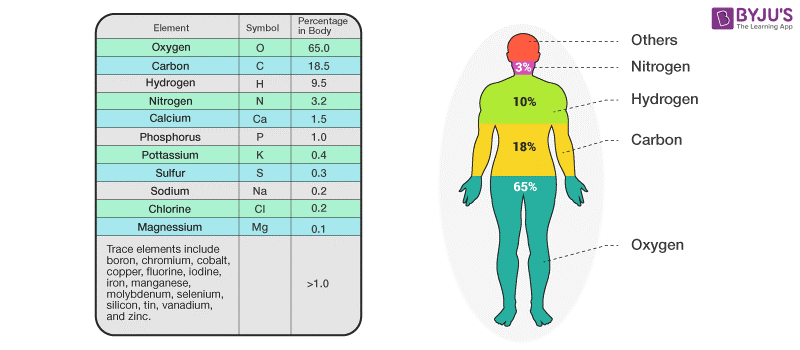
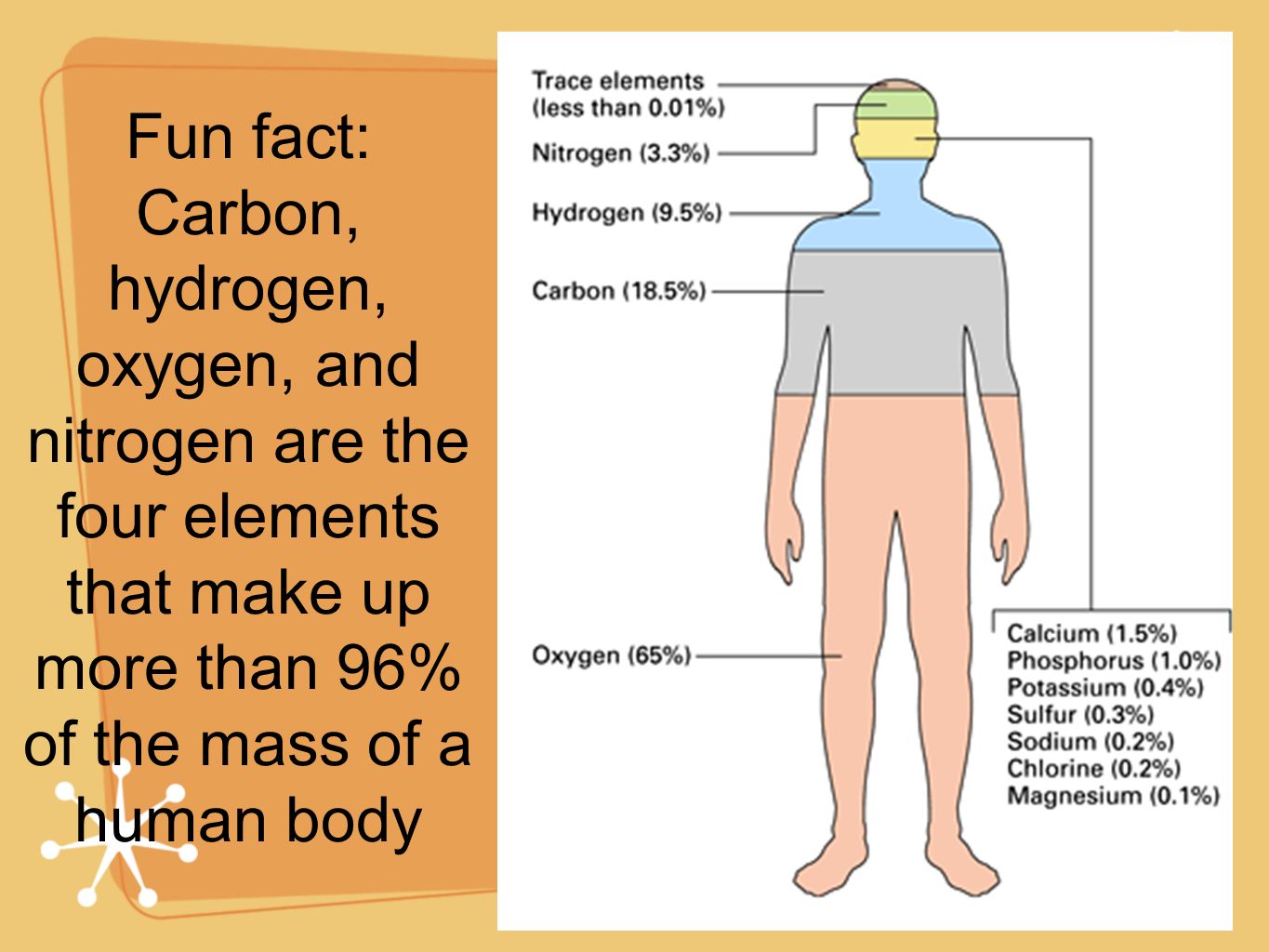
There are 92 naturally occurring elements on Earth, from hydrogen to uranium, so why do these four make up such a majority of living things? Part of the reason lies in the fact that they are versatile elements, capable of producing a vast array of chemical compounds, but it also has to do with the fact that they are among the most abundant elements in the universe. Together they make up about 96% of our bodies, as you can see in the figure above. These four atomic elements are oxygen, carbon, hydrogen, and nitrogen. One of the things we have learned is that we-and every other living thing on Earth-are made up mostly of four elements. Over the past two centuries, we have gained a much better understanding of the atomic elements and how they have formed. ASU School of Life Sciences The abundance of elements in the human body. The four elements also connected to the four humours of the human body, which formed a basis of Western medicine until the 1800s. It was a central aspect of alchemy until Robert Boyle demonstrated there were more than four elements in 1661. The idea that everything was made of these fundamental elements had a deep influence on early Western science. These were not elements in the modern sense, but rather essences that gave everything their physical properties. Plato referred to them as the four elements. Around 450 BC, the Greek philosopher Empedocles wrote that the world was comprised of four things: earth, air, fire and water.
